Introduction
Spina bifida is one of the most common birth defects worldwide. This is a defect in the closure of the vertebral column that may or may not involve the spinal cord or meninges (covering layers of central nervous system) or both being out-pouched. SB is called a neural tube defect (NTD).
Types of SB
There are three types of SB,
- Spina bifida occulta (surface area of some of the vertebrae is not completely closed).
- Spina bifida cystica with meningocele (meninges are pushed into the gaps of vertebrae).
- Spina bifida cystica with myelomeningocele (meninges and spinal cord protrude through the opening).
Treatment
SB’s complications are managed according to the problem. In hydrocephalus ( where CSF accumulates in the cavities of the brain) a shunt is used to drain off the surplus fluid. When the problem is related to elimination functions catheterization and vesicostomy are employed where one cannot pass urine due to bladder dysfunction.
The protruded part of the meninges and spinal cord out of the body is treated with two types of surgery. One is pre-natal surgery and the second is post-natal surgery. But there is still no treatment for the damaged part of the central nervous system. Studies and researches are being done to discover how to repair the damaged part of central nervous system. One of them is stem cell treatment.
Introduction of stem cell treatment
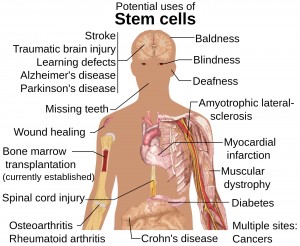
Treatment by stem cells is a new way of treating the neural tube birth defects. Stem cells are undifferentiated pluripotent cells which are known to be a type of cells that have tendency to differentiate into any type of cells. These cells are present in the fetus and differentiate into cells for all of the organ systems. Thus they differentiate into heart cells, brain cells, muscle cells , bone cells and so forth . Because of their characteristics, stem cells, are being used to repair damaged parts of the body. In SB, a part of spinal cord is protruded and damaged. This damage causes many complications, such as disability, bladder and cognitive problems as the cells of central nervous system don’t have dividing ability. Damaged cells of the spinal cord in spina bifida do not repair themselves. But, the stem cells have the ability to divide and to differentiate into cells of the spinal cord. When stem cells are introduced in a damaged area, they can repair cells of that area. So, complications due to damaged cells can be minimized.
Stem cells (Source: www.1001.nccdn.net)
Source of stem cell
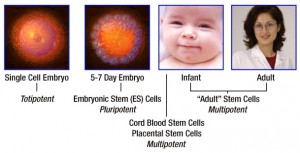
Figure 1. Stem cells can be extracted from pre-natal as well as post-natal patients. In pre-natal patients, embryonic cells, amniotic mesenchymal stem cells and umbilical cord blood are collected for their stem cells [1]. In post-natal patients, stem cells can be collected from bone marrow and fat cells. In stem cell treatment, stem cells are collected from the patient to minimize the immunity response against these cells.
Figure 2. Multipotent stem cells (Source: www.1001.nccdn.net)
Spina Bifida Stem cell treatment methods
Two methods of stem cell treatment are being used these days.
- One method of stem cell treatment is that stem cells are introduced during pregnancy. Pre-natal surgery can minimize the damage to the spinal cord. These results can be obtained when surgery is performed before 20-25 weeks of pregnancy. Pre-natal surgery during this period can abort pregnancy. Pre-natal surgery after that period is useless because damage to the spinal cord is prominent at this stage. Pre-natal stem cell treatment is performed 10-12 weeks before the pre-natal surgery period. Stem cells are introduced into the womb.
- Another method of stem cell treatment is performed after the delivery. In this method, stem cells are introduced by three ways: 1) by intravenous injection, in this condition stem cells must cross the blood-brain barrier to reach the central nervous system, 2) by intrathecally by a lumbar puncture. The blood-brain barrier is not as closed in younger patients compared to older ones.
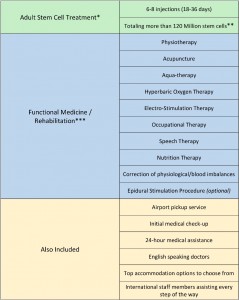
Figure 3 below gives the promotion for a stem cell treatment programme in China designed for adults.
Figure 3. Beike Biotechnology stem cell treatment strategy in China
(Source: www.stemcelltreatmentnow.com)
Benefits of stem cell treatment for spina bifida
To date the result of stem cell therapy for spina bifida is extremely encouraging. Stem cell treatment is a new way of treating birth defects. Many serious birth defects can now be treated by stem cells. Spinal cord injury is treated well with stem cell treatment. Spina bifida treatment by stem cells is performed at many medical centers. But, this is still at the research level. Stem cell treatments do create many improvements in sensation and muscle supplies of bowel/bladder and legs as well as in cognitive and intellectual performance. Stem cell treatment is also being performed for bowel or bladder incontinence, commonly associated with SB.
Side effects
Stem cell treatment is still in the trail stage. At some centers, it is still performed in animals so that we may know the long term side effects this newly developing technique [3]. There is a fear that it can cause a brain tumor. Stem cells are pluripotent so these cells can differentiate to any type of cell or tissue. Sometimes, they differentiate to other type of cells instead of repairing damaged neurons. Sometimes stem cells can cause urinary infection and other urinary complications when intestinal stem cells are used in connection with bladder or bowel incontinence.
Caution
Stem cell treatment is still in the trail and experimental stage. So a 100% result is not assured. So, the patient may face some long term side effects. There is often much improvement, but results are not predictable with stem cell treatment. This is an experimental and expensive method of treatment.
Hospitals with stem cell treatment facilities
- Nova Cells Institute Mexico: Phone: (1-562-916-3410), E-mail: NCinfodesk@gmail.com
- Regenerative Medicine Institute in Mexico phone: (1.866.668.9263)(1.619.621.2800).
- Beike Biotechnology, Better Being Hospital: Address: East Block, 2nd Floor, Yuanxing Technology Building, #1 Songpingshan Road, Nanshan District, Shenzhen, Guangdong, China 518057.
References
1. Turner CG, Klein JD, Wang J, Thakor D, Benedict D, Ahmed A, et al. The amniotic fluid as a source of neural stem cells in the setting of experimental neural tube defects. Stem Cell Dev 2013;15;22(4):548-53.
2. Li H., Gao F, Ma L, Jiang J, Miao J, jiang M, et al. Therapeutic potential of in utero mesenchymal stem cell (MSCs) transplantation in rat foetuses with SB aperta. J Cell Mol Med 2012;16(7):1606-17.
3. Baker KS, Bresters D, Sande JE. The burden of cure: long-term side effects following hematopoietic stem cell transplantation (HSCT) in children. Pediatr Clin North Am 2010;57(1):323-42.